 Nuclear fission reactions produce incredibly large amounts of energy compared to chemical reactions. Material that can sustain a nuclear fission chain reaction is said to be fissile or fissionable. For instance, when one mole of U-235 undergoes fission, the products weigh about 0.2 grams less than the reactants this lost mass is converted into a very large amount of energy, about 1.8 × × 10 10 kJ per mole of U-235. If this process continues, a nuclear chain reaction occurs. For fission of Uranium-235 the most common radioactive fission products include isotopes of iodine, cesium, strontium, xenon and barium. 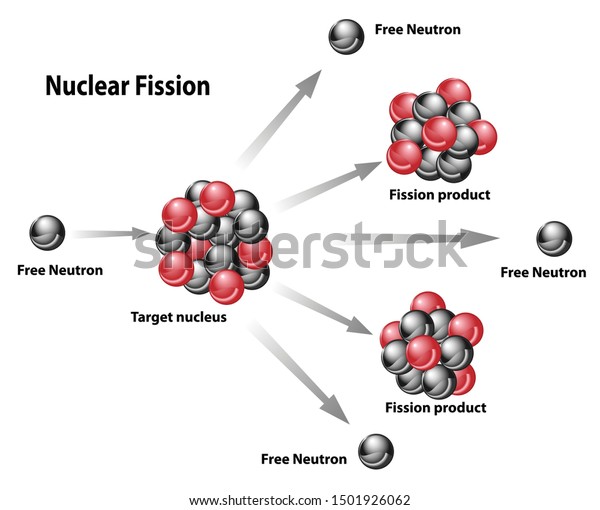
You could make this a focus of your coverage of this area: the physics ideas won’t tell you what’s. Contribution to the study of fission products release from nuclear fuels in severe accident conditions: effect of the pO2 on Cs, Mo and Ba speciation. In words this would be: A uranium-235 atom absorbs a neutron to become uranium-236 which then undergoes fission to form the products xenon-140 and strontium-93. Students may have strong feelings about nuclear technology power stations and bombs. In contrast, refractory peak-yield fission products. 1) and are known to fractionate severely from Pu, making reliable in-terpretation difficult (5, 15). Radioactive decay of both fission products and transuranic. 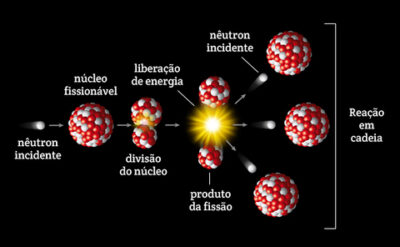
\): The fission of a large nucleus, such as U-235, produces two or three neutrons, each of which is capable of causing fission of another nucleus by the reactions shown. This topic does not lend itself to much practical work there are a few analogue demonstrations which can be valuable. Although long-lived fission products such as 137Cs and 90Sr are detectable in Trinity nuclear debris today (914), these radio-nuclides have volatile precursors (e.g., 137I and 137Xe Fig. Nuclear reactors work by containing and controlling the physical process of nuclear fission. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
